Prologue
When a flowering branch placed in a vase continues to thrive longer than expected, it is often thanks to a florist’s special touch–her secret ingredient, a dash of salt dissolved carefully into the water. Surprisingly, this is not a modern discovery but a hidden botanical wisdom first revealed over a century ago by the botanist Wilhelm Pfeffer and later explained intuitively by Jacobus H. van’t Hoff, for which he was awarded the first Nobel Prize in Chemistry in 1901. Throughout this series, we’ll highlight pivotal, award-winning breakthroughs that have profoundly shaped the course of chemistry over time.
What’s Going on Within the Bottle or Vase?
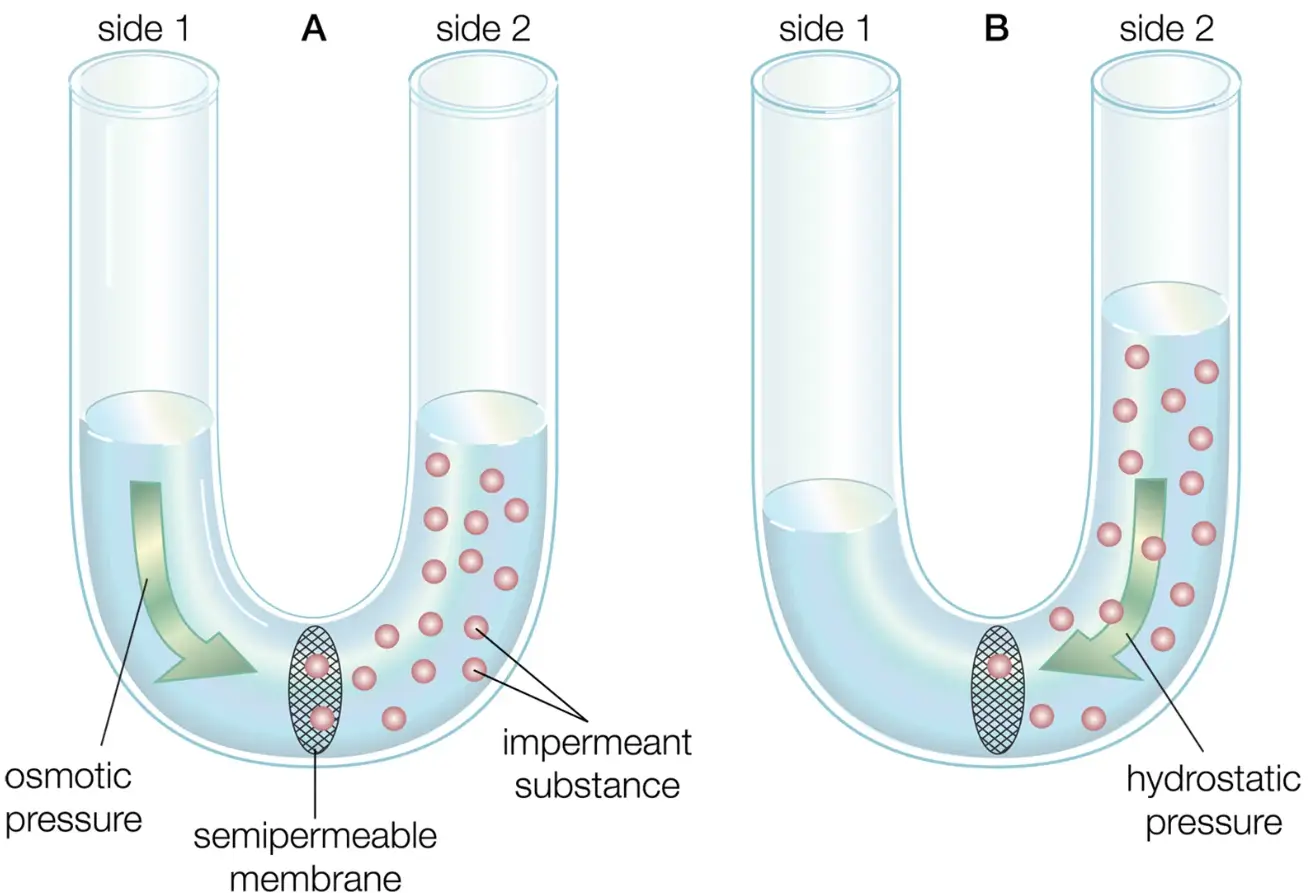
What exactly is happening inside the vase or the bottle? This botanical magic is explained by a phenomenon known as osmotic pressure. Imagine placing a membrane between two liquids: pure water on one side and a sugar-water solution on the other. The membrane allows water molecules to pass through easily but blocks larger sugar molecules. Driven by natural forces seeking balance, water molecules move across this barrier into the sugar solution, creating an increased pressure on the solution side. This pressure, pushing from within, is called osmotic pressure.
Interestingly, all these remarkable visualizations emerged at a time when even the concept of molecules was still somewhat uncertain. This speaks volumes about the exceptional creativity and scientific imagination of Jacobus H. van’t Hoff.
It’s not just florists who benefit from this fascinating natural process. Osmotic pressure is also the hidden mechanism behind the mighty oak tree’s ability to transport sap upwards, reaching even its highest branches. The tree uses osmotic pressure to draw water upward from its roots, defying gravity to nourish its farthest leaves.
Let us look a bit more in detail and see how osmotic pressure would also lead to the wilting of flowers in the vase. Flowers stay fresh when water flows easily into their cells. Osmotic pressure helps draw water into the cells, keeping them firm and upright. When flowers are placed in a vase with water, initially they absorb water through their stems, and their cells become full, keeping the flowers fresh and standing upright. But over time, as the water in the vase becomes less pure (due to dissolved substances like sugars, bacteria, or other impurities), the osmotic pressure outside the flower cells changes. Water is then less likely to move into the cells, and in some cases, water actually moves out of the flower cells into the vase water. As a result, the flower cells lose water and shrink, causing the flowers to become soft and droopy. This shrinking and softening is what we call “wilting.” In short: When impurities build up in the vase water, osmotic pressure decreases the amount of water entering the flower cells, causing them to lose firmness and wilt.
Thus, this invisible force of osmotic pressure, once a florist’s secret, connects the simple beauty of a flower in a vase to its wilting to the awe-inspiring majesty of towering trees.

van’t Hoff intuitively thought that this osmotic pressure should be somehow related to the pressure of a gas. So let us now look at his window of thought and compare osmotic pressure with the pressure of a gas. However, we need to keep in mind that osmotic pressure and gas pressure are two distinct phenomena related to different physical and chemical properties of matter. Although both involve the concept of pressure, they originate from fundamentally different processes and conditions.
On Osmotic Pressure and Pressure in General
Osmotic pressure is defined as the pressure required to stop the flow of water across a semipermeable membrane from a region of low solute concentration (more dilute) to a region of higher solute concentration. It arises due to the presence of dissolved particles or solutes in a solution. Water tends to move spontaneously to equalize concentrations on both sides of a membrane. However, this movement can be counteracted by applying external pressure, and the pressure necessary to halt water movement entirely is called osmotic pressure. This phenomenon typically occurs in solutions that are separated by membranes, such as in biological cells, plant roots, and dialysis equipment. For instance, plant cells rely on osmotic pressure to draw water into their interior, which maintains their firmness and structural integrity. The wilting of plants or flowers happens because osmotic pressure inside the cells drops, causing water to leave the cells, resulting in loss of rigidity and visible wilting.
Several factors influence osmotic pressure. It primarily depends upon solute concentration — the more concentrated the solution, the higher its osmotic pressure. It also depends on temperature; raising temperature usually increases osmotic pressure. Osmotic pressure is described mathematically by the van ‘t Hoff equation ($pi$ = iMRT), where $pi$ represents osmotic pressure, i the van ‘t Hoff factor (number of particles produced per dissolved molecule), M the molar concentration, R the gas constant, and T the temperature in Kelvin.
On the other hand, gas pressure is defined as the force exerted by gas molecules colliding continuously with the walls of their container. Unlike osmotic pressure, which is driven by differences in concentration across membranes, gas pressure results purely from random molecular motion and collisions. Gas molecules are in constant rapid movement and collide frequently with container surfaces, producing pressure evenly in all directions.
Gas pressure commonly occurs in everyday scenarios, such as inflating balloons, car tires, or using gas cylinders. It is directly influenced by three main factors: the number of gas particles (moles of gas), temperature, and container volume. Increasing the number of gas molecules or raising the temperature boosts gas pressure, while increasing the container’s volume typically reduces it. Gas pressure is mathematically defined by the Ideal Gas Law (PV = nRT), in which P is gas pressure, V is volume, n represents moles of gas, R is the universal gas constant, and T is temperature in Kelvin.
Directionally, osmotic pressure acts through a membrane, pulling water toward a region of greater solute concentration. In contrast, gas pressure acts outward uniformly, pressing equally against all surfaces of its container. Thus, osmotic pressure has a directional nature, whereas gas pressure does not — it is isotropic, meaning it is the same in all directions.
In summary, while both osmotic pressure and gas pressure measure how particles exert force, osmotic pressure relates specifically to water movement due to solute concentration gradients across a semipermeable membrane. Gas pressure, however, involves free-moving gas molecules colliding against container surfaces. Understanding these fundamental differences helps clarify their distinct roles and importance in chemical and biological processes. We now would like to find out when we ‘squeeze’ a solution what happens to the osmotic compressibility. Likewise, we will also ask what happens when we squeeze a gas, what happens to the gaseous compressibility. Now we leave a thought-provoking question here: Can these compressibilities give a hint on the structure of a solution or a gas? We will visit this question later. But first, let us look at osmotic compressibility and compressibility of a gas to appreciate the genius of van’t Hoff even more.
Osmotic pressure and osmotic compressibility, along with gas pressure and gas compressibility, are concepts related to how easily solutions or gases change volume when external conditions, such as pressure or concentration, change.

Of Osmotic and Gas Compressibility
Now, osmotic compressibility relates to how much the volume of a solution changes when the osmotic pressure changes. In simple terms, it tells us how easily a solution can be “squeezed” or “expanded” by changing the concentration of dissolved substances. If a solution is highly compressible, a small change in osmotic pressure will greatly affect its volume. Conversely, if it has low compressibility, it resists volume change even when osmotic pressure varies. This is important in biological systems, where maintaining the right water balance in cells is crucial. Cells must keep their volume fairly stable, so they have relatively low osmotic compressibility, allowing them to resist large volume changes.

The relationship between osmotic pressure and osmotic compressibility is straightforward: the higher the osmotic pressure, usually caused by a higher concentration of dissolved substances, the more difficult it becomes to compress the solution further. A very concentrated solution typically has lower compressibility, meaning its volume won’t easily decrease even if more solute or pressure is added.
Next, let’s look at gas pressure and gas compressibility. Gas pressure results from gas molecules bouncing around and hitting the walls of their container. If you fill air into a balloon, the molecules collide with the inside walls, creating pressure and causing the balloon to expand.
Gas compressibility measures how easily the volume of a gas changes when pressure is applied. Gases are usually very compressible. If you squeeze a gas into a smaller space, its volume shrinks noticeably because gas molecules are widely spaced and can move closer together easily. For example, air pumped into a bicycle tire becomes compressed, increasing its pressure and reducing its volume significantly.
The relation between gas pressure and gas compressibility is very direct and predictable. When pressure on a gas increases (for example, when you compress air into a small tank), the gas volume decreases significantly. Conversely, reducing the pressure allows the gas to expand and occupy more space. This predictable relationship is described by Boyle’s Law, stating that gas pressure and volume are inversely related: as one increases, the other decreases.
In summary, osmotic pressure and osmotic compressibility deal with solutions and how concentration changes affect their volumes, whereas gas pressure and gas compressibility deal with gases and how easily their volumes change with external pressure. Solutions typically have lower compressibility, resisting changes in volume, while gases have high compressibility and respond readily to changes in pressure. Does this tell us anything about the internal structure of a solution or a gas? Indeed, it does. The above observation implies that a solution is less compressible and has less void in itself than a gas. The structure of a gas has more empty space than that of a solution or a liquid. But there is more to that. In fact, the Boyle’s law which we referred to earlier along with the equation for osmotic pressure refers to ideal gases and ideal solutions. But real-life situations are more complex. There is non-ideality involved in real gases and real solutions. In fact, in 1869, Andrew’s isotherms were published which details how the real gases change volume with pressure and temperature. It was even observed in the isotherm that gases change phase, meaning they liquefy and thus shedding more light on their structure. Van’t Hoff very nicely captured this liquefaction with non-ideality and proposed the structure of real gases in a more mathematical way. Much later the same logic was used to glean structure of liquids from osmotic pressure in the same way as structure of gas was gleaned by van’t Hoff, which is popularly known as van’t Hoff equation for real gases.
We will now look very simply how osmotic pressure, osmotic compressibility and criticality of phases and gas pressure, gaseous compressibility and phase transitions in gases relate with the idea and if we can arrive at the structure of a liquid from osmotic compressibility.
Let us have a quick recap. Osmotic Pressure is simply the pressure needed to prevent water from flowing from an area with fewer dissolved particles (solutes) into an area with more dissolved particles through a thin barrier called a semipermeable membrane. Imagine two containers separated by a thin film that allows only water, not solutes, to pass through. If one side has pure water and the other has saltwater, water naturally moves toward the salty side to balance concentrations. To stop this movement, you must apply pressure on the salty side. The exact amount of pressure required to stop this flow is the osmotic pressure.
This process is fundamental to biology. For example, plant cells use osmotic pressure to draw water in, keeping them firm and upright. If osmotic pressure drops, cells lose water and wilt. Similarly, when you sprinkle salt on cucumber slices, water comes out because the salt creates a high osmotic pressure outside, pulling water from inside the cucumber.
Osmotic Compressibility is a related concept but slightly different. Compressibility measures how much the volume of something (like a liquid or a solution) changes when pressure changes. Osmotic compressibility specifically describes how easily the volume of a solution can change due to changes in osmotic pressure or solute concentration.
If something has high osmotic compressibility, its volume easily changes when osmotic pressure changes. For example, a dilute solution (like weak saltwater) is usually more compressible because small changes in solute concentration cause noticeable changes in volume. A very concentrated solution (like very salty water) is usually less compressible because its structure is tightly packed, resisting volume changes.
On Criticality, Phase transitions and Structure of Liquid
Now we look at the criticality of phases. Criticality of Phases refers to special conditions (called critical points) where differences between phases (gas, liquid, solid) disappear. Normally, water can exist as ice (solid), liquid water, or steam (gas). Each phase is clearly different. However, if you heat water and increase pressure simultaneously, eventually you reach a point called the critical point. At this critical point, the distinction between liquid water and steam disappears. Above this critical point, water is neither purely liquid nor purely gas — it’s a single “supercritical fluid.”
Criticality is important because at this point, physical properties (like density or compressibility) become unusual — small changes in pressure or temperature dramatically change the substance’s properties. At critical conditions, compressibility becomes very high, meaning even a tiny pressure change greatly affects volume. Now we look back again at gas pressure and gas compressibility.
Gas Pressure is caused by countless gas molecules bouncing around and hitting the walls of a container. Imagine pumping air into a balloon. Gas molecules collide with the inside walls, pushing them outward. The more molecules (or the faster they move due to heating), the higher the gas pressure becomes. Gas pressure, thus, depends on three simple things: the number of gas molecules, temperature, and container volume.
Gas Compressibility describes how easily gases can change their volume under pressure. Gases are highly compressible because gas molecules are far apart, freely moving, and can be squeezed closer together. For instance, when you pump air into a bicycle tire, you compress the gas molecules into a smaller volume. As you compress the gas, pressure quickly increases because more molecules hit the walls more frequently.
This behavior is well described by Boyle’s Law: increasing pressure shrinks the volume of a gas proportionally, showing that gases have high compressibility compared to liquids and solids. Now we discuss what happens when these phases like gas, liquid, solid change, which is known as phase transitions.
Phase Transitions are changes between states of matter — solid, liquid, and gas. For gases, common phase transitions include condensation (gas becoming liquid) and vaporization (liquid becoming gas). These transitions depend strongly on temperature and pressure.
Imagine water vapor (steam) in a container. If you cool it slightly or increase the pressure, the gas molecules slow down, move closer together, and begin sticking together to form liquid droplets. This transition from gas to liquid is condensation. Conversely, heating a liquid increases the energy of molecules, causing them to break free and become gas, called vaporization or boiling.
Near phase transitions, gases show noticeable changes in compressibility. For instance, near condensation points, gas compressibility increases sharply because molecules become easier to squeeze together into a liquid.
Now we correlate osmotic pressure, osmotic compressibility, gas pressure and gas compressibility and try to arrive conceptually at the structure of liquids.
Osmotic pressure and compressibility in solutions are conceptually similar to gas pressure and compressibility in gases but with some fundamental differences. Both involve particles moving and responding to pressure or concentration changes. Osmotic pressure arises from concentration differences across a membrane, while gas pressure comes from free molecular collisions in open space.
At critical conditions — where gases become supercritical fluids — the compressibility of the fluid sharply rises, somewhat resembling how solutions react to changes in osmotic pressure. At critical points, fluids become extremely sensitive to small changes, similar to dilute solutions experiencing small volume changes due to osmotic pressure shifts. Thus, observing these changes at critical points, we are able to get an idea on the structure of the liquid. Thus, the question arises, can we derive the structure of a liquid from Osmotic Compressibility?
Indeed, osmotic compressibility can provide insights into the structure of liquids, especially solutions. When studying osmotic compressibility, scientists indirectly understand how tightly molecules or particles are packed together. If a solution is highly compressible, it means molecules or particles are loosely packed or flexible, changing easily with external conditions like pressure or osmotic pressure. Conversely, low osmotic compressibility means particles are packed tightly, forming a structured, less flexible liquid.
By measuring osmotic compressibility across various concentrations and temperatures, we learn how molecules interact and arrange themselves in solutions. This gives clues about liquid structure. For example, if adding a small amount of salt significantly reduces compressibility, it implies that the salt ions strongly influence water structure, creating organized, structured clusters around themselves. Thus, from measuring osmotic compressibility, scientists can indirectly determine how structured or disorganized a liquid is at the molecular level. This helps explain why some liquids flow easily, while others resist changes, maintaining shape and volume.
By closely examining osmotic pressure and compressibility, you can ingeniously craft your fruity yogurt, ensuring that its particles stay magnificently apart — never clumping, never sticking, resulting in the creamiest, smoothest yogurt imaginable. Similarly, by diving deeply into the magic of osmotic pressure, you can engineer a diaper so extraordinarily effective that the little one sleeps peacefully, blissfully unaware of even the faintest trace of moisture touching her delicate skin. The study of osmotic pressure marches on triumphantly, empowering scientists to invent astonishing new materials — from brilliantly durable paints to irresistibly smooth yogurts and decadently perfect ice creams. Next time you watch a flower droop, savor an unusually delightful yogurt, or taste that impeccably velvety ice cream, give a playful mental nod to van’t Hoff — the visionary who kickstarted this incredible journey over a century ago, whose legacy continues to shape the pleasures and comforts of everyday life even today. That’s the joyful message behind it all.
Epilogue
As we have seen, osmotic pressure is far more than a florist’s trick — it’s a fundamental force that shapes life from the drooping petals in your vase to the mighty ascent of sap in ancient oaks. Van’t Hoff’s insight — that a solution’s hidden pressures mirror those of a gas — opened a window onto the microscopic architecture of liquids long before molecules were fully understood. Today, that same principle guides innovations as diverse as silky yogurts and ultra-dry diapers, reminding us that even the simplest observations can yield the most trans formative technologies.
By comparing osmotic and gas compressibility, and exploring their dramatic swings at critical points, we edge closer to decoding liquid structure itself. Every experiment, every equation, brings us a step nearer to understanding how particles dance, cluster, and propagate the unseen pressures that sustain life. So next time you marvel at a wilting bloom or sip a perfectly smooth ice cream, remember: you’re witnessing the elegant legacy of osmotic pressure — a constant, invisible partner in the ever-unfolding story of matter. We will see some more captivating phenomena in the next article of this series.
References
[1] Wikipedia contributors, Jacobus Henricus van’t Hoff — Wikipedia, The Free Encyclopedia, [Online; accessed 10-September-2025]. [Online]. Available: https://en.wikipedia.org/w/index.php?title=Jacobus_Henricus_van_%27t_Hoff&oldid=1305919539.
[2] Foflonker, Fatima, “Osmotic pressure,” in Encyclopedia Britannica. May 2025.
Further Reading
The works presented here are not directly referenced in the text and are intended for informational purposes only.
[3] J. H. Van’t Hoff, “Osmotic Pressure of Solutions,” Zeitschrift für Physikalische Chemie, pp. 481–508, 1887.
[4] T. Andrews, “Researches in Steam at High Pressures,” Philosophical Transactions of the Royal Society of London, pp. 575-590, 1869.
[5] P. Atkins, J. De Paula, and J. Keeler, Atkins’ Physical Chemistry, en, 11th ed. London, England: Oxford University Press, Dec. 2017, Chapters 5 (Solutions) and 7 (Gases).
[6] B. E. Poling, J. M. Prausnitz, and J. P. Connell, The Properties of Gases and Liquids. New York, NY: McGraw-Hill, 2001, Chapters 1–3. Definitive reference on fluid properties, covering compressibility factors, the ideal gas law vs. real-fluid corrections, and detailed data for both gases and solutions.