Prologue
Long before we invented the vocabulary of nanoscience, before crystallographers learned to see atoms arranged like constellations, and before chemists dared to imagine hollow crystals capable of breathing, hosting, and transforming molecules — there existed a curious idea in the mind of an ancient philosopher.
Chuang Tsu once wrote that the true usefulness of a pot lies not in its clay walls, but in the empty space within.
More than two thousand years later, a young Japanese student sat with this thought open before him: the usefulness of uselessness. The book that held it — The World of Genius — was written by Nobel Laureate Hideki Yukawa. But it was Chuang Tsu’s whisper from antiquity that stayed with him.
Years later, that student would grow into Susumu Kitagawa, a pioneer who dared to chase what other researchers dismissed as trivial: porous structures made from metals and organic molecules. The world would come to know them as Metal Organic Frameworks — MOFs for short — one of the most versatile materials ever engineered. But when Kitagawa began, porous organic materials were considered unstable, impossible, even foolish.
Everything important, people believed, must be dense. Density meant stability. Density meant utility. Density was chemistry.
But what if, Kitagawa wondered, the space between the atoms mattered as much as the atoms themselves?
From that question began the quiet revolution of MOFs, one that now shapes energy research, climate solutions, clean air, drug delivery, hydrogen storage, water harvesting, and even the philosophy of modern materials science.
This is the story of MOFs — not as a technical review, but as a human saga of curiosity, perseverance, imagination, and the remarkable chemistry of empty space.
And like all good stories, it does not start in the laboratory.
It starts with a question.
The Idea that Stood Alone
In the 1980s, solid-state chemists were racing toward materials that behaved like metals: dense organic conductors, magnetic organic crystals, complex inorganic lattices. The dream was clear — build solids whose charge, spin, or optical properties could rival the robustness of metal oxides.
Porosity? That was the opposite of progress. To most researchers, pores were chemical weaknesses — voids that would collapse, sites of instability, imperfections to eliminate.
But to Kitagawa, trained in coordination chemistry yet guided by the philosophy of curiosity, porous structures were not defects. They were opportunities. If organic molecules could link to metal ions to create strong open networks, then the resulting materials could host guests, recognize shapes, separate molecules, tune reactions, and even change behavior depending on what entered their internal world.
He saw a framework, not a flaw.
He envisioned a space that does chemistry, not simply contains it. Yet in those early years, almost no one believed him. The community favored thermodynamically stable dense materials. Reviewers rejected proposals. Conference attendees doubted his data. He recounts attending a Gordon Conference where, after presenting his results, colleagues questioned him relentlessly—even during breakfast the following morning.
But he did not stop. He kept assembling metal ions with organic linkers. He kept building structures that held together. He kept exploring the “usefulness of uselessness.” And then, he found it. A stable, crystalline, permanently porous material held together by coordination bonds — the early form of what we now call a Metal – Organic Framework. The world had not yet understood it. But chemistry had quietly changed.
What Exactly is a MOF?
To appreciate the story fully, we must first step gently into the world of MOFs.
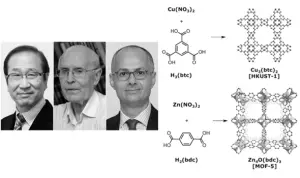
A Metal – Organic Framework is, at its heart, a marriage between two very different families of matter:
- Metal nodes – atoms or clusters such as zinc, copper, zirconium, cobalt, or rare-earth ions.
- Organic linkers – molecules containing oxygen, nitrogen, or sulfur atoms that can bind to metals, often shaped like rigid rods or branching connectors.
When these two components self-assemble, they form crystalline frameworks resembling scaffolds, lattices, or molecular architecture. What makes MOFs extraordinary is not merely their structure but the fact that the spaces inside them — the pores — are uniform, adjustable, and addressable at the atomic scale.
Here is why they matter:
- Enormous Surface Area: One gram of a typical MOF can have a surface area larger than an entire football field. This means molecules entering the MOF have countless places to stick, react, assemble, or transform.
- Tunable Functionality: Change the metal. Change the organic linker. Change the geometry. The properties shift like an artist switching brushes.
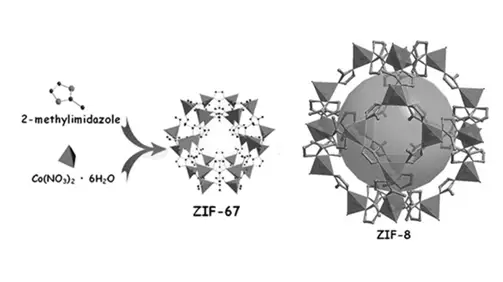
- Modular Design: MOFs assemble like molecular LEGO blocks. By choosing different building units, one can make: hydrophobic pores, hydrophilic pores, catalytic sites, flexible channels, rigid cages, fluorescent frameworks, magnetic structures and so on. This “modular chemistry” makes MOFs one of the most designable classes of materials known today.
- Applications Spanning Entire Industries: They are used or envisioned in: CO2 capture, water harvesting in deserts, hydrogen storage, drug delivery, separation of gases, detoxification of nerve agents, catalysis for fine chemicals, electrochemical energy storage, sensors for pollution, smart membranes, even materials that bend, contract, or soften when exposed to stimuli.
In essence, MOFs are functional spaces — chemically active rooms at the nanoscale. They do not merely contain chemistry. They are chemistry.
The Four Generations of a Revolution
Through decades of scientific evolution, MOFs have gone through distinct eras — each like a movement in an unfolding symphony. Kitagawa himself classifies them into four generations, each born from deeper understanding and bolder creativity.
- Generation 1: Frameworks Without Breath: The earliest coordination polymers lacked permanent porosity. They collapsed when guests left. They were beautiful but fragile — like soap bubbles without elasticity.
- Generation 2: True MOFs with Permanent Porosity: A breakthrough occurred when robust organic–metal structures retained their shape even when evacuated. This made them comparable to zeolites but far more versatile. This was the moment the world began to pay attention. MOFs had arrived.
- Generation 3: Flexible MOFs or Soft Porous Crystals: Here the story becomes even more magical. Some MOFs behaved like breathing organisms — they expanded or contracted depending on what entered their pores. Their structures were flexible yet crystalline. They responded to gases, temperature, light, or pressure. Kitagawa predicted such flexibility in 1998 — long before it became experimentally celebrated. Soon, the world recognized soft porous crystals as a rich new arena: materials that change shape without losing identity.
- Generation 4: Defects, Disorder, Liquids, and Hybrids: This is the frontier of MOF science today. The newest generation embraces: hierarchical structures, hybrid organic–inorganic composites, intentional defects, amorphous or liquid-state MOFs, anisotropic pores, phase transitions between solid, liquid, and glass-like states.
In this era, the chemistry of MOFs is no longer about building perfect crystals. It is about engineering the imperfections to unlock new behaviors. The story of MOFs, then, mirrors the evolution of scientific thinking: from rigidity to flexibility, from order to useful disorder, from static materials to dynamic ones that live, breathe, and respond.
A Journey of Persistence: The Human Story
Behind every scientific idea stands a human being — a person who faces doubts, failures, rejections, and long nights when nothing seems to make sense. Kitagawa’s journey is a testament to perseverance. For years, he could secure only small grants — enough to support minimal experiments. Many reviewers rejected his proposals. Students were few. Facilities were limited. He recalls commuting on a slow train each day, using that one hour to revise student manuscripts. He gave feedback the very next day, no matter how busy he was.
He trained generations of young researchers not only through instruction but through example. He spoke to them as equals, encouraged speeches at laboratory gatherings, and cultivated independence. He believed curiosity — not obedience — was the true currency of good research. In those difficult years, he never stopped. At age 46, he published the first robust MOF structure. At age 51, he published in Science, proving the ordered arrangement of oxygen molecules inside MOF channels. This was a landmark: the first time such crystallographically resolved gas arrays were observed in a porous organic framework. After that, the world could not ignore MOFs.
Global researchers entered the field. Grants flowed. Applications exploded. The chemistry of emptiness had become a global frontier. His story teaches us something profound:
Revolutions in science do not always begin with brilliance. They begin with stubbornness.
The Beauty of Space: An Intuitive Look at Why MOFs Matter
Let us pause from biography and enter imagination. Picture a vast hotel, stretching beyond sight, filled with identical rooms. Each room has furniture carefully arranged to attract specific types of guests. Now shrink that hotel until it fits on the tip of a needle. You are now imagining a MOF.
A MOF may choose to allow carbon dioxide in but exclude water vapor. Or admit xenon atoms but reject nitrogen. Or host a catalyst that performs a reaction only when confined within its nano-chambers. Every pore is a decision. Every channel is a pathway. Every atom inside the lattice plays a quiet but essential role.
Some MOFs behave like living creatures: They inhale gases and exhale them later. They swell when heated. They twist when illuminated. They soften when hydrated. They stiffen when pressurized. This “softness” is not weakness—it is functionality.
MOFs let chemists dream like architects. Want a framework that changes color when exposed to explosives? You can design it. Need a structure that traps greenhouse gases with extraordinary efficiency? You can build it. Want to store hydrogen at safe pressures for clean-energy vehicles?
MOFs are among our best hopes.
This is why MOFs are one of the fastest-growing fields in chemistry today: because they exist at the intersection of imagination and molecular precision.
MOFs and the Grand Challenges of our Time
To appreciate the impact of MOFs, imagine the biggest problems humanity faces:
- Can we capture CO2 efficiently?
- Can we purify water sustainably?
- Can we store hydrogen safely and densely for clean energy systems?
- Can we separate industrial gases without enormous energy costs?
- Can we sense toxins in air or water before they harm us?
- Can we deliver drugs to specific target sites inside the body?
- Can we harvest moisture from desert air?
- Can we catalyze reactions with fewer emissions and less waste?
MOFs offer paths to each of these questions. Let us explore a few.
Capturing Carbon Dioxide
Carbon capture is often energy-intensive because gases must be cooled, compressed, or chemically reacted. MOFs offer: high surface area, tunable binding sites, selective adsorption of CO2 over N2, regenerability with minimal energy, and artificial photosynthesis, which has even been done in my research group.
Some MOFs can bind CO2 as gently as Velcro — strong enough to hold, weak enough to release without excessive heating. In global decarbonization pathways, MOFs appear repeatedly: in industrial chimneys, natural gas purification, direct air capture, and even artificial leaves.
Storing Hydrogen
Hydrogen is the cleanest fuel imaginable — its combustion produces only water. But storing hydrogen safely is a grand challenge. MOFs can store hydrogen by adsorption rather than compression, potentially reducing the need for ultra-high-pressure tanks. The uniform nano-pockets of MOFs can hold hydrogen molecules like eggs in a carton, increasing density while enhancing safety.
Separating Industrial Gases
Chemical industries spend more than 10% of global energy on separations alone — distilling, filtering, and splitting mixtures. MOFs offer precision separation without heating:
- ethanol/water separation,
- CO2/CH4 separation,
- olefin/paraffin separation,
- oxygen/nitrogen separation.
This is transformative. It can reduce carbon emissions by gigatons and even convert CO2 into useful fuels like formic acid, which is a hydrogen carrier.
Water Purification and Desalination
Certain MOFs attract water molecules and funnel them through pores small enough to block contaminants. Some can even harvest atmospheric moisture — a dream for drought-prone regions. One MOF called MOF-303 demonstrated remarkable ability to extract water from desert air using only sunlight.
Catalysis Inside a Confined World
Reactions inside MOF pores behave differently from reactions in open solution. Confinement can: stabilize transition states, orient molecules, enhance selectivity, accelerate reaction pathways, and create unusual products. Catalysis inside MOFs is like cooking inside a pressure cooker — the environment changes the outcome.
MOFs in Medicine
Imagine a framework that carries a drug through the bloodstream, protecting it from degradation, and only releases it when it reaches a tumor. Or consider a MOF that binds harmful toxins in the body. The porous, customizable, biocompatible nature of certain MOFs makes them ideal for: imaging, drug delivery, radiotherapy enhancement, antimicrobial activity, and biosensing. The field is young but promising.
In the words of another Nobel laureate, Professor Omar Yaghi’s student, Professor Rahul Banerjee of IISER Kolkata,
Metal–Organic Frameworks are a remarkable class of materials with applications ranging from direct air capture to water harvesting. I had the privilege of working with their inventor, Professor Omar Yaghi, and I am convinced that these materials will fundamentally transform the way we live.
The MOF Universe: Flexibility, Disorder, and the New Frontiers
The most exciting developments today come from embracing imperfections. Intentionally introducing missing linkers or metals creates sites that: enhance gas uptake, improve catalytic activity, increase conductivity, and create gradients inside crystals. This is defect engineering — a paradox where the best materials are no longer perfect.
MOFs can host nanoparticles, polymers, or biomolecules inside their pores. These composites can perform multiple functions at once, like Swiss-Army-knife materials.
The idea that a crystalline MOF can melt and become a liquid — and then solidify into a glassy MOF — is groundbreaking. It blurs the boundary between organic and inorganic matter.
Such materials could be: moldable, processable, dynamic, and compatible with electronic devices.
Nature builds porous structures everywhere: proteins, enzymes, bone, shells. MOFs are beginning to emulate these designs, creating enzyme-like catalysts or bio-inspired recognition systems. The future of MOFs is not merely scientific. It is artistic.
MOFs as a Philosophy of Science
MOFs teach us something elemental about thinking itself. The scientific world often celebrates what is visible: atoms, bonds, numbers, formulas. But MOFs remind us to look at what is invisible: the empty spaces, the unoccupied volumes, the channels through which possibilities move, the architectures of absence, the chemistry of the void.
The idea that emptiness can be functional, that the absence of matter can generate new forms of matter, is a profoundly philosophical notion — one that resonates with Taoism, Zen, and even the patterns of nature.
A honeycomb is strong not because of wax, but because of its geometric voids. A building stands because of rooms, not walls. A flute makes music because of its hollowness. The universe itself is largely dark and unseen.
MOFs, in their essence, are scientific expressions of this truth. They are the chemistry of possibility.
The Lessons of Kitagawa: Science as a Human Endeavor
As we reflect again on Kitagawa’s journey, we encounter several lessons that transcend chemistry: He pursued a question that others ignored. He valued imagination over immediate utility. He trusted the usefulness of the seemingly useless. Years passed before the world appreciated his work. He persisted through skepticism, rejections, and slow progress. He kept going even when no one else believed. He built small but determined groups of students. He led through kindness, mentorship, and flat hierarchy. He praised publicly, guided privately, and encouraged independence. He did not chase fashionable topics. He asked fundamental questions: What happens inside space? How does confinement shape chemistry?
This vision guided decades of discovery.
His breakthrough Science paper came from collaborating with physicists. MOFs today touch biology, physics, materials science, energy engineering, and environmental science. The frameworks he built were not just chemical — they were intellectual frameworks for partnership. His journey tells us: Science grows where imagination meets perseverance; where curiosity meets courage.
A World Transformed by MOFs
We now live at a time when MOFs are woven into the scientific landscape. Yet their future is even brighter. MOFs may become essential to carbon capture at power plants, cement factories, and even in air-cleaning devices. Their tunability allows unprecedented efficiency. Hydrogen storage and delivery systems using MOFs could accelerate the hydrogen economy. MOFs also appear in batteries, supercapacitors, and catalysts for clean fuels like those for the reduction of CO2. Highly selective MOFs can remove heavy metals, purify industrial effluents, and sense pollutants at extremely low concentrations. Atmospheric water harvesters using MOFs may bring affordable, clean water to arid deserts and remote communities. Conductive MOFs, once considered impossible, are emerging for sensors, electrodes, and optoelectronic devices. From targeted drug delivery to diagnostic imaging, MOFs have begun entering biomedical engineering. Then there are MOFs beyond our imagination. Soft MOFs that bend like rubber. Liquid MOFs that flow like honey. MOF membranes that filter at the molecular level. MOFs with magnetic, luminescent, catalytic, or switchable properties. We stand at the edge of a vast new universe — one built not of planets and stars but of atoms arranged to create functional emptiness.
A Story of Science and Humanity
Let us return to the beginning of this story—the moment a student read about Chuang Tsu’s pot, the pot whose usefulness lies in emptiness. That idea, once philosophical, became chemical. It became structural. It became technological. It became a global research field. But more importantly, it became a metaphor for the scientific spirit itself.
Science is not merely the accumulation of facts. It is the cultivation of spaces—spaces where imagination can breathe, where doubt can live, where curiosity can grow, where young minds can wander.
In a MOF crystal, space is designed to host molecules. In a research laboratory, space must be created to host ideas. Kitagawa’s story teaches us that the void is not a vacancy. It is an invitation.
An invitation to think differently. To assemble old components in new ways. To see potential where others see emptiness. To trust the quiet voice of curiosity. To persevere when misunderstood. To build frameworks—not just of matter, but of meaning.
Epilogue: The Future Belongs to the Open Mind
As we close this journey, let us imagine the future of MOFs not as a destination but as an unfolding horizon. A young scientist somewhere today may be reading an old philosophical text, just as Kitagawa once did. Something in it may spark a question so subtle that the world ignores it — at first.
That question may lead to a new kind of framework. A new kind of material. A new way of thinking about space, matter, motion, or energy. A new chapter in the story of science. The lesson from MOFs is simple:
- Do not fear the empty spaces.
- They are where creativity begins.
- They are where possibility lives.
- They are where the future is built.
And in the great framework of progress, every pore — every curiosity, every doubt, every question — has a purpose.
Further Reading
- H. Furukawa, K. E. Cordova, M. Okeeffe, and O. M. Yaghi, “The chemistry and applications of Metal-Organic frameworks,” Science, 2013.
- O. M. Yaghi, M. Okeeffe, N. W. Ockwig, H. K. Chae, M. Eddaoudi, and J. Kim, “Reticular synthesis and the design of new materials,” Nature, vol. 423, pp. 705–714, 2003.
- H. Li, M. Eddaoudi, M. Okeeffe, and O. M. Yaghi, “Design and synthesis of an exceptionally stable and highly porous Metal-Organic framework,” Nature, vol. 402, pp. 276–279, 1999.
- B. Liu, “Interview with professor Susumu Kitagawa,” ACS Mater. Lett., vol. 1, pp. 564–567, 2019.


I enjoyed reading this article. It covers a topic that I had no knowledge of and didn’t initially think I would find this interesting.
But the author, who is clearly an expert in the area of MOFs, has presented this topic in such a compelling and fascinating style that I was totally impressed. I also found the list of real-life applications of MOFs in solving some complex problems very interesting.
Thank you very much for your kind comments. I am glad that you enjoyed reading the article.